Nobel Prize for Medicine 2018 for the immunotherapy of tumors: the comment of Prof. Bonini
In the 90s, separately, two scientists identified different molecules that function as a brake on the immune system, preventing it from recognizing and attacking cancer cells: thanks to this discovery, James P. Allison and Tasuku Honjo won the Nobel Prize for Medicine o Physiology 2018. Allison has opened the way to this research by studying proteins that make the immune system “fall asleep” and sensing their great potential: by manipulating their natural brake would have been possible to attack tumors with new weapons. Honjo contributed by discovering a protein on immune cells and eventually revealed that it also operates as a brake, but with a different mechanism of action.
Both findings have translated over time into new approaches to cancer therapy that proved to be strikingly effective in the fight against cancer.
We discussed this with Chiara Bonini, Professor of Hematology at UniSR and Head of the Experimental Hematology Unit of the IRCCS San Raffaele Hospital.
Prof. Bonini, is this Nobel important?
Yes, it definitely is: it is an important Nobel, fair, that gives hope. The Nobel Prize for Medicine must give hope, and this one in particular does so, because it rewards the discovery, choosing two great scientists that represent it, of the mechanisms through which the cancer cells “restrain” the immune system and make sure that this does not see the tumor, and therefore cannot fight it. Thanks to the studies of these researchers, we have dramatically enriched our knowledge of these mechanisms: to know them means that we can also modify them, by removing the “brake” to the immune system and unleashing this natural weapon to recognize and attack cancer cells. It is a Nobel Prize awarded to tumor immunotherapy [which studies how to “exploit” the immune system to fight cancer, Editor’s note].
Of all the researchers involved in this field, why were Allison and Honjo selected?
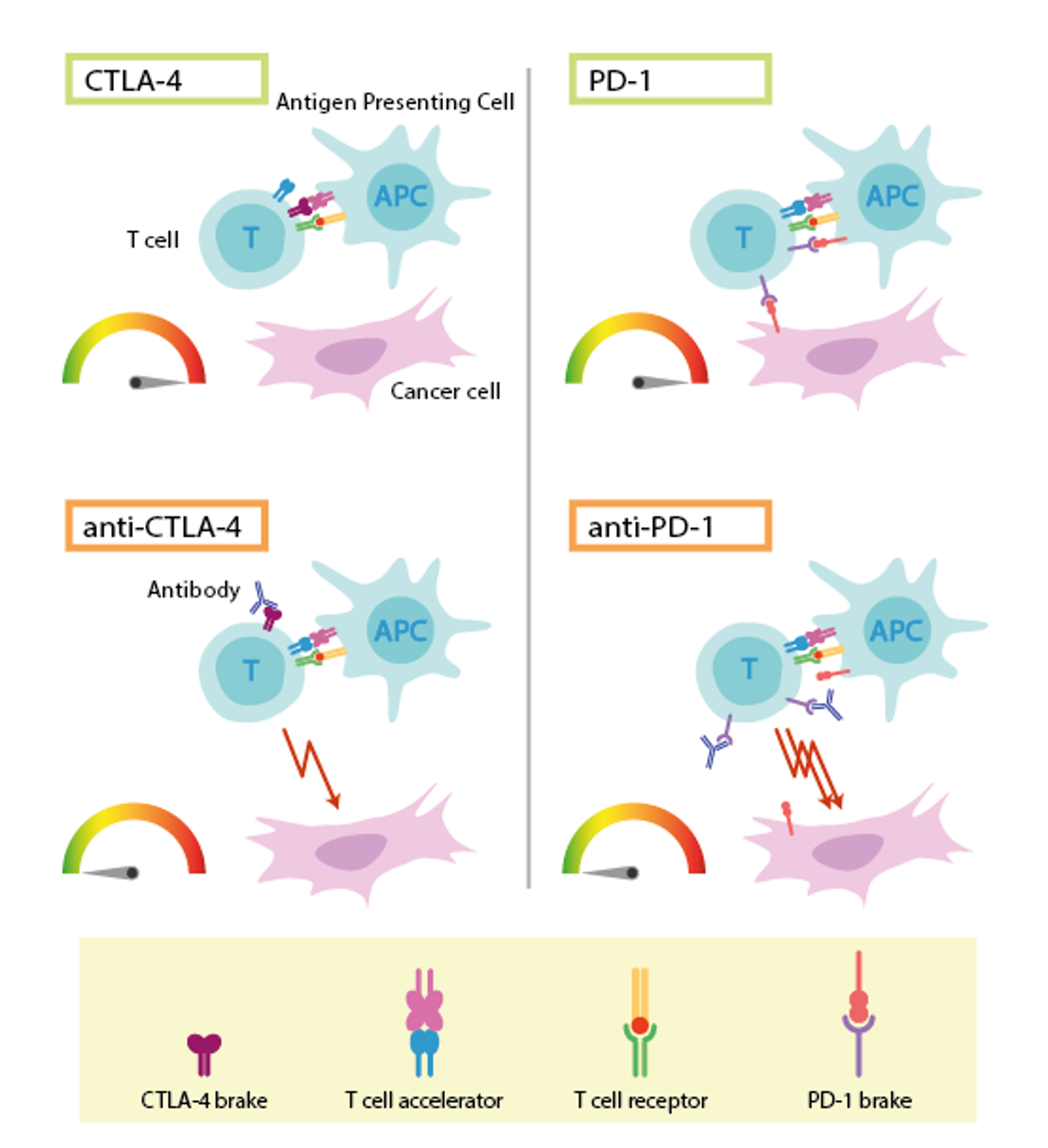
Because they were the first to discover the two molecules that prevent the immune system from the in tumor recognition: CTLA-4, discovered by Allison in 1991, and PD-1, identified by Honjo a year later. Such molecules are expressed by T lymphocytes (cells of the immune system) when they deplete their function; the tumor exploits this feature to inhibit the lymphocyte by binding to these receptors. “Masking” these molecules with antibodies (anti-CTLA-4 and anti-PD-1) makes it sure that the lymphocytes are no longer inhibited, and therefore are able to recognize and attack the tumor. CTLA-4 and PD-1 are still the most exploited molecules from a clinical point of view, in recognition of the value and importance of this discovery that unquestionably deserves the Nobel Prize.
Upper left: Activation of T cells requires that the T-cell receptor binds to structures on other immune cells recognized as ”non-self”. A protein functioning as a T-cell accelerator is also required for T cell activation. CTLA- 4 functions as a brake on T cells that inhibits the function of the accelerator. Lower left: Antibodies (blue) against CTLA-4 block the function of the brake leading to activation of T cells and attack on cancer cells. Upper right: PD-1 is another T-cell brake that inhibits T-cell activation. Lower right: Antibodies against PD-1 inhibit the function of the brake leading to activation of T cells and highly efficient attack on cancer cells. Picture modified from: © Mattias Karlén / The Nobel Committee for Physiology or Medicine.
Tumor immunotherapy: where are we in San Raffaele?
In San Raffaele we have an enthusiastic and very well-coordinated community of researchers dealing with tumor immunotherapy. The wealth of skills of our researchers allows us to study, in a very detailed way, the multiple and complex aspects of the encounter between the tumor cell and the immune system. Clinically, San Raffaele is a center of excellence that sees very high numbers of patients affected by different kinds of tumor, and that manages them with great care at every stage, from diagnosis to the therapeutic path.
In my opinion, the important aspect is that in our Institute the assistance and research activities combine perfectly. The presence of a hospital, with doctors who attract and visit patients in all the stages of different diseases, is a true benefit for the research, because it translates into a contribution of information, samples and the possibility of studying these pathologies always better. The result is a virtuous circle that gives credibility and visibility to the Institute, which in this way becomes a reference center for the patient who needs treatment according to the best guidelines, but also – if necessary – with the last experimental drug.
What is like working and studying at San Raffaele?
Working at San Raffaele is a privilege. The new drugs, especially those deriving from this kind of research, are very expensive, in their preclinical preparation, but especially in their production. San Raffaele remains an almost miraculous Institute, in a difficult country that invests very little in research. As for being a student at the Vita-Salute San Raffaele University, I think we should ask them directly; what is certain is that the aspects that I spoke about – among which the close and profitable collaboration between research and clinic – are at hand, ready to be analyzed and embraced by our students.
On the left, James P. Allison; on the right, Tasuku Honjo. Author: Niklas Elmehed. © Nobel Media

Tags:
You might be interested in

QS Rankings by Subject 2026

UniSR ranked 1st in Italy for Research and Teaching
/resolutions/res-c660x528/Veschetti_Cariplo_P.-Aeruginosa_UniSR-(1).jpg)
Uncovering the hidden role of bacterial microRNAs in chronic respiratory diseases