The ongoing COVID-19 pandemic has forced the scientific community to devote itself to the research and development of various strategies to fight SARS-CoV-2 infection. The only informed way to identify new antivirals is through the knowledge of the three-dimensional structure, literally the shape, of the virus proteins that are responsible for cell infection and virus replication. Once the structure of proteins is known, it is possible for chemists to synthesize compounds that block its function, and consequently the virus life cycle.
We talk about it in this cycle of 5 episodes with the help of Dr. Massimo Degano, Group leader of the Biocrystallography Unit of the IRCCS San Raffaele Hospital and lecturer at our University (he teaches Chemistry at the Degree Course in Dentistry and Dental Implantology and at the International MD Program, Biochemistry at the Degree Course in Medicine, and Structural Biology at the Degree Course in Biotechnology Research in Medicine).
What is Coronavirus Spike Protein and how does it work?
One of the most interesting virus target proteins for this purpose is the Spike (S) protein. This protein decorates the surface of the virus by forming characteristic protuberances (making it look like a crown – hence the name “Coronavirus”). Protein S is composed of two parts:
- S1, which contains a region that serves to bind to the target cell by adhering to the human ACE2 protein, that serves as the virus receptor;
- S2, which in a second phase allows the virus to enter the cell.
Hence, a molecule that can block the interaction between the Spike protein and ACE2 would potentially be able to prevent coronavirus infection and, consequently, the disease.
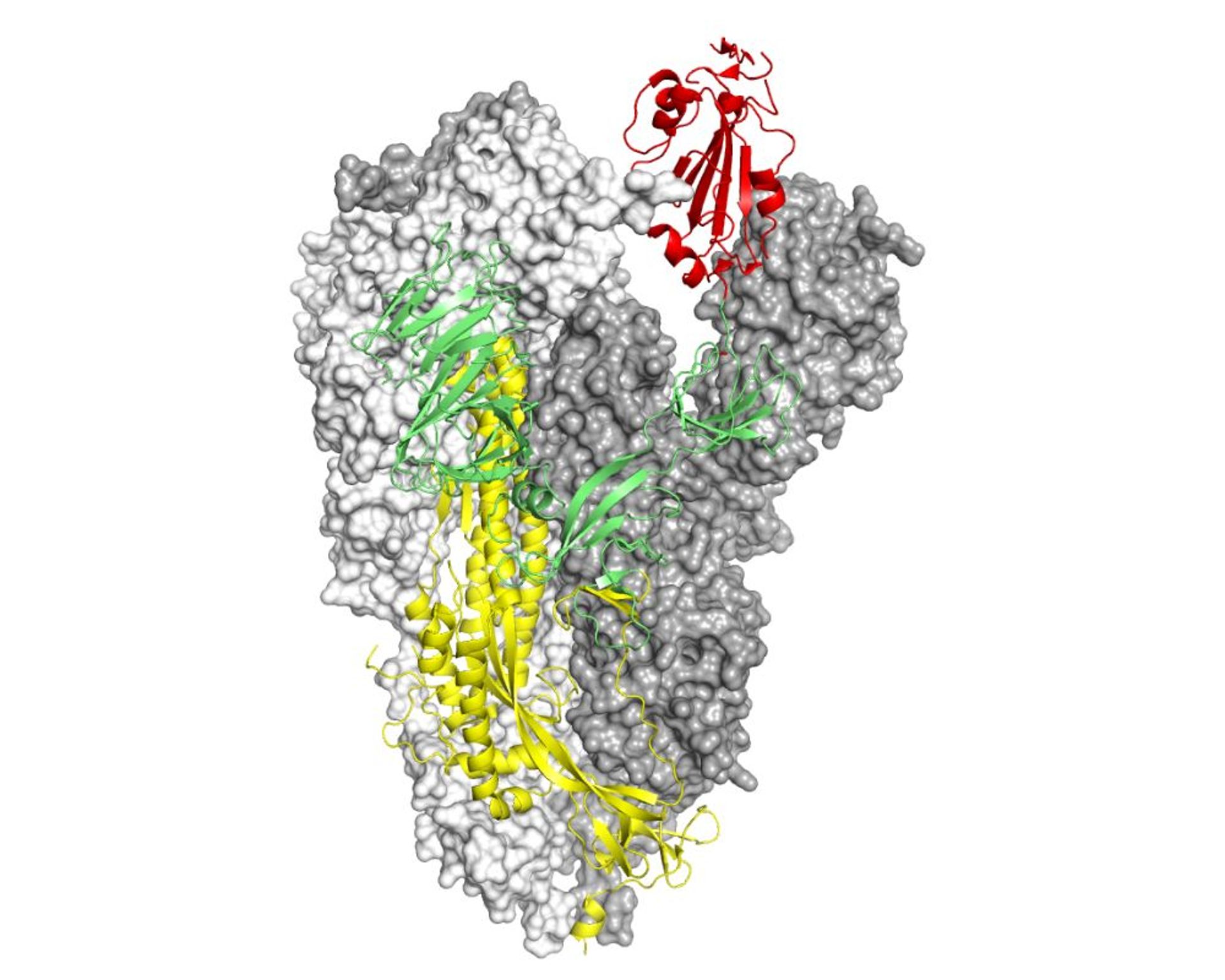
Structure of the Spike protein from SARS-CoV-2. Two chains are represented through the molecular surface which highlights the general shape of the molecule. The S1 chain is colored in green, with the portion that serves to interact with the ACE2 receptor to infect cells in red. The S2 chain, containing the part of the molecule necessary for the virus to enter the cell, is colored yellow. Kind courtesy of Dr. Degano.
What is the purpose of visualizing its structure?
Why is seeing the structure of a protein essential for developing new drugs? Imagine having to unscrew a bolt and not have the wrenches available: if you see the shape of the bolt and its dimensions you will be able to forge (or choose) the correct size wrench to succeed in your purpose. Similarly, when the structure of a protein is visible, the molecule can be engineered with the size and shape that can optimally bind to it and block its function.
There are essentially two techniques that allow us to visualize protein structures and to describe them in great detail: X-ray crystallography and cryo-electron microscopy (cryo-EM).
- In X-ray crystallography, the protein molecules are arranged according to an ordered and symmetrical lattice, and when they are irradiated by a very intense beam of X-rays they in turn emit radiation that can be measured: through computer calculations, they can tell us exactly how the electrons are distributed around the atoms in the crystal (the electron density);
- in cryo-EM, single molecules are irradiated with electrons that through highly sophisticated lenses are focused to similarly give us the image of electronic density. Thanks to this information, viewable with graphics programs on PCs, for structural biologists it is a question of solving a puzzle by inserting the amino acids that make up the protein into the electron density and reconstructing the structure of the protein.
Spike and RBD-ACE2
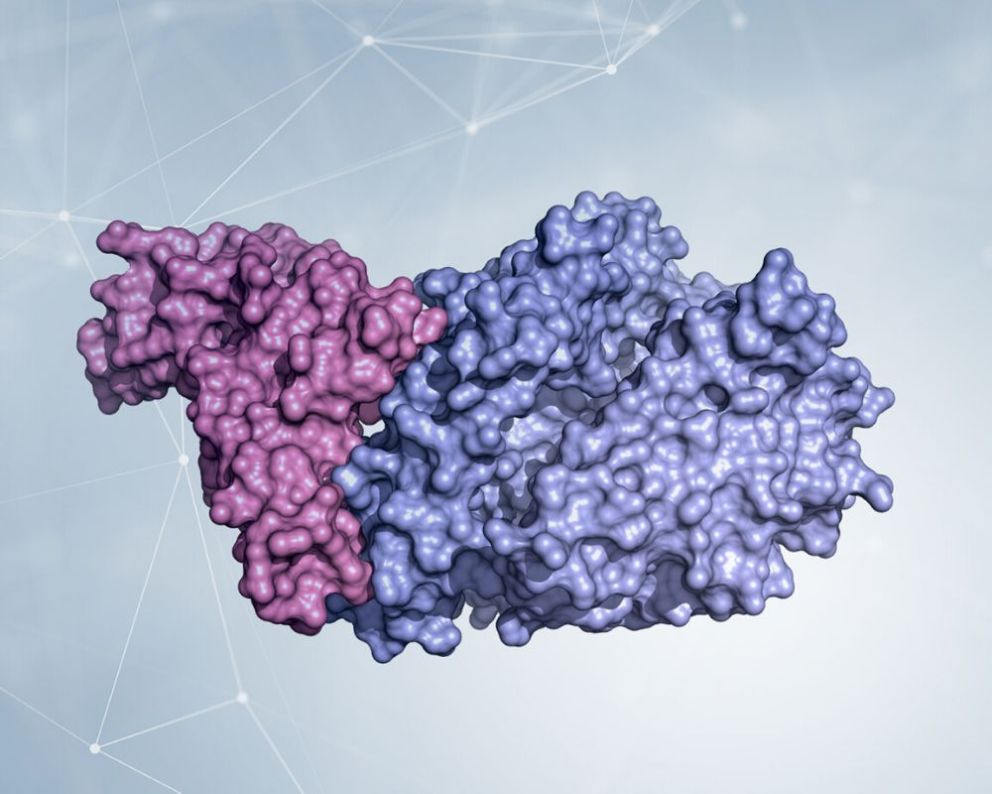
Within two months from the first cases of COVID-19, two research groups independently determined the structure of the Spike protein using cryo-EM [1],[2], showing that it is made up of three identical chains associated (this is called a “trimeric” arrangement) and consists of a region that resembles the stem of a flower with the region essential for contact with the cells to be infected (called RBD, receptor-binding domain) in place of the petals. This part of the molecule is flexible like a wind vane in the wind, and can “search” for ACE2 molecules with which to interact. Subsequently, the structure of ACE2 linked to Spike [3] was visualized by crystallography, showing in very high detail how the virus attaches to the cell to be infected.
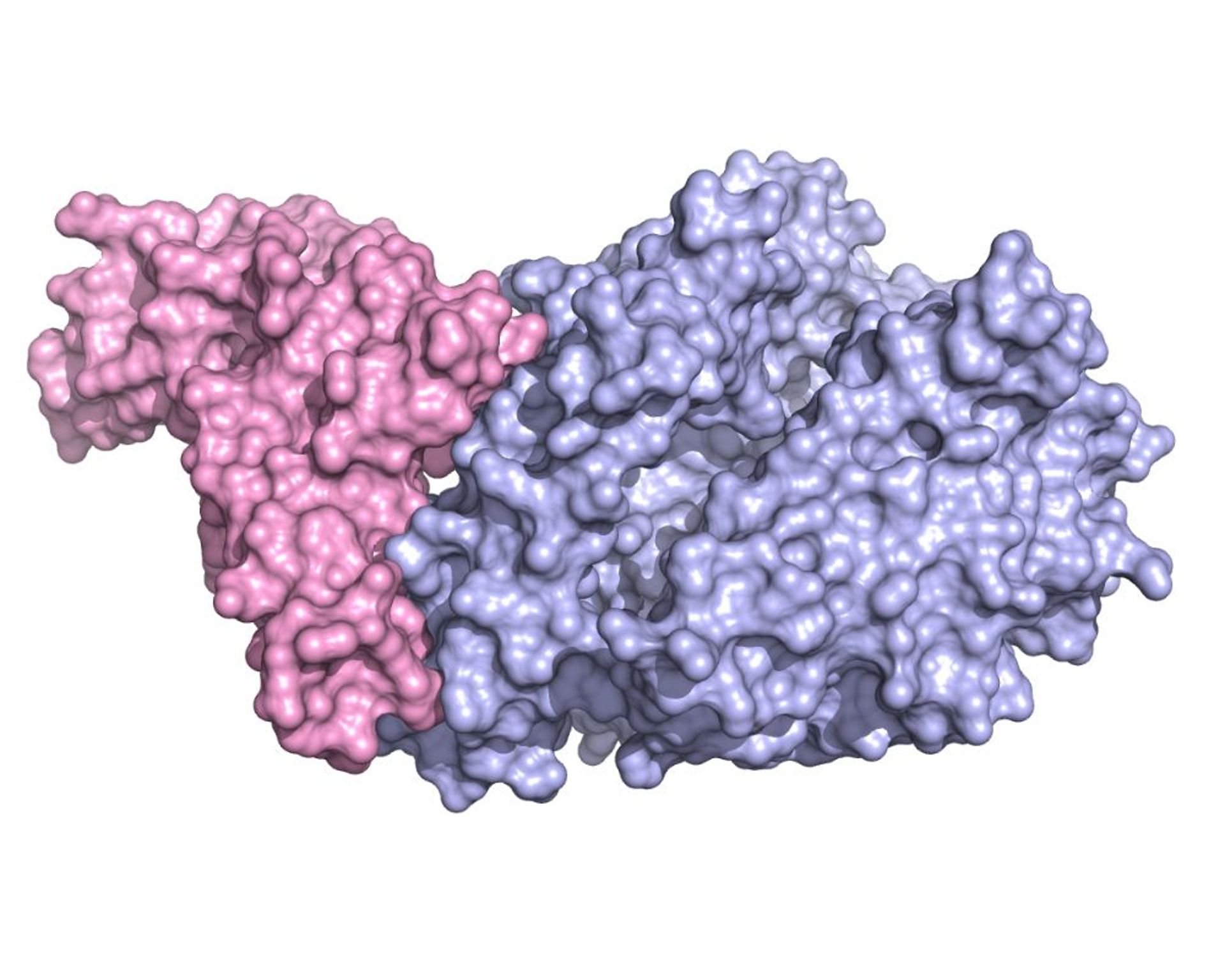
The crystallographic structure of the domain that binds Spike’s receptor (RBD, colored in pink) combined with the extracellular part of the enzyme ACE2 (in light blue) shows in detail which parts of the two molecules are involved in the adhesion between virus and cell. Kind courtesy of Dr. Degano.
The challenge now is to use this knowledge to find molecules capable of binding to Spike, keeping busy the part that should interact with ACE2, and thus preventing infection.
Read the other episodes of the series "Structural biology for SARS-CoV-2"
Second episode: "Cryo-electron microscopy: small electrons to visualize large molecules"
Third episode: "Developing antivirals in a rational way: “blunting” the SARS-CoV-2 scissors"
Fourth episode: "Structural biology for SARS-CoV-2: X-ray crystallography"
Fifth episode: "RNA polymerase, the SARS-CoV-2 “sloppy photocopier”"
References
[1] D Wrapp et al. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science 367, 1260-1263 (2020). DOI: https://doi.org/10.1126/science.abb2507
[2] AC Walls et al. Structure, function and antigenicity of the SARS-CoV-2- Spike glycoprotein. Cell 181, 281-292 (2020). DOI: https://doi.org/10.1016/j.cell.2020.02.058
[3] J Shang et al. Structural basis of receptor recognition by SARS-CoV-2. Nature 581, 221-224 (2020). DOI: https://doi.org/10.1038/s41586-020-2179-y
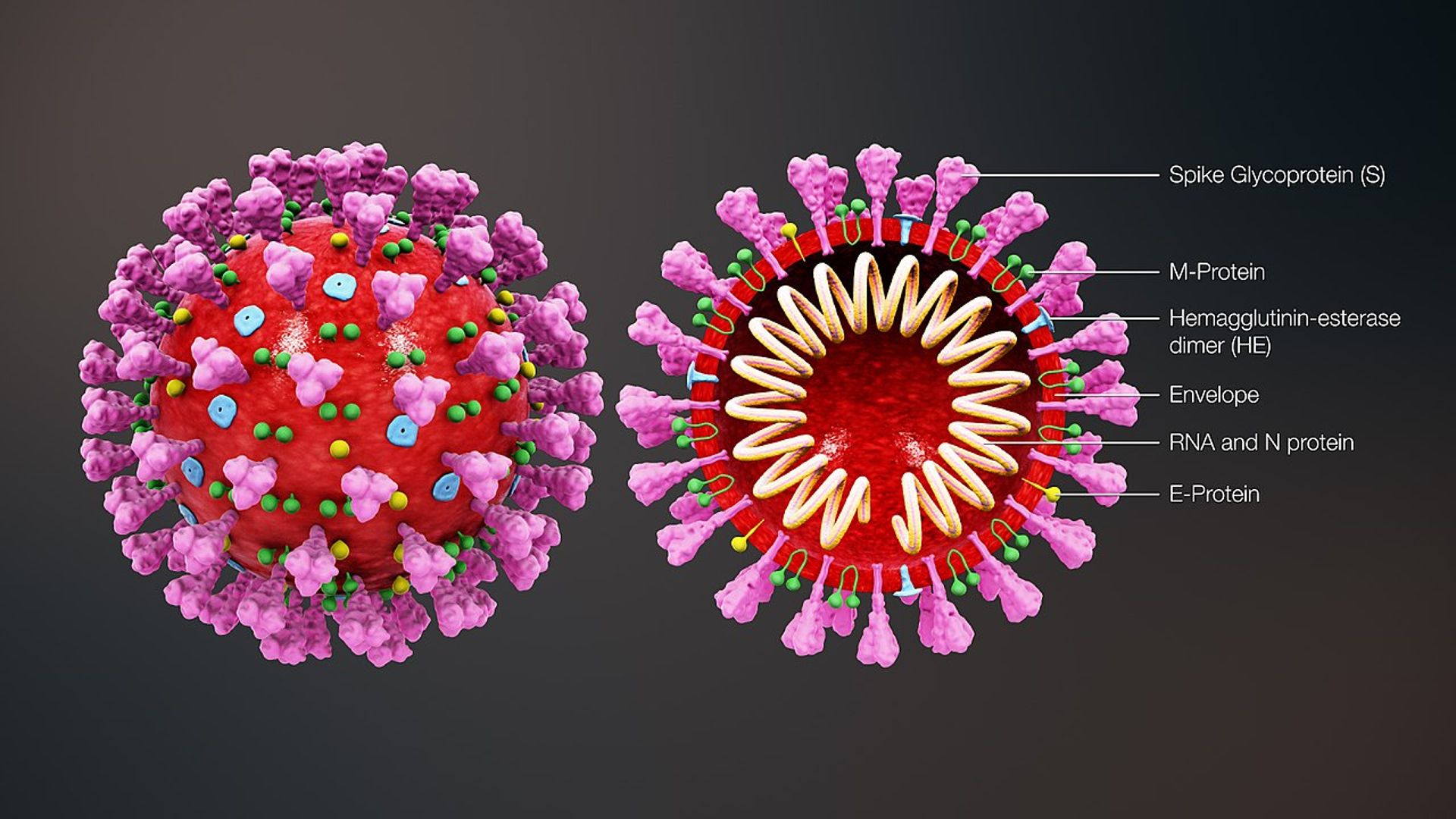
Text image: illustration of a SARS-CoV-2 virion (the single viral infectious particle), complete and in section (credit: https://www.scientificanimations.com/wiki-images/).
You might be interested in

UniSR designated as WHO tuberculosis research centre: what we will do together

World Rare Disease Day: Research at UniSR Continues All Year Round

Multiple sclerosis, breakthrough in research: researchers have identified a molecule that promotes repair of the nervous system