Thanks to a funding from Fondazione Cariplo for young researchers, it was possible to study the mechanisms underlying thrombus formation in patients undergoing LVAD implantation. The project manager is Filippo Consolo, Biomedical Engineer and Researcher at the Faculty of Medicine and Surgery UniSR.

After a degree in Biomedical Engineering at the Polytechnic of Milan and a PhD in Bioengineering from the Polytechnic of Turin, he spent a long period in the United States (Dept. Biomedical Engineering, Stony Brook University), Portugal (Dept. of Physics, Faculdade de Ciencias e Tecnologia) and Bologna (Dept. of Chemistry Università Alma Mater Studiorum). He then returned to PoliMi where he spent 6 years as a PostDoc, during which he worked on experimental bio-fluid dynamics, bioreactoristics and microfluidics. In 2016 he was the winner of the Fondazione Cariplo “Biomedical research conducted by young researchers” funding, which allowed him to start an itinerary as a researcher in UniSR.
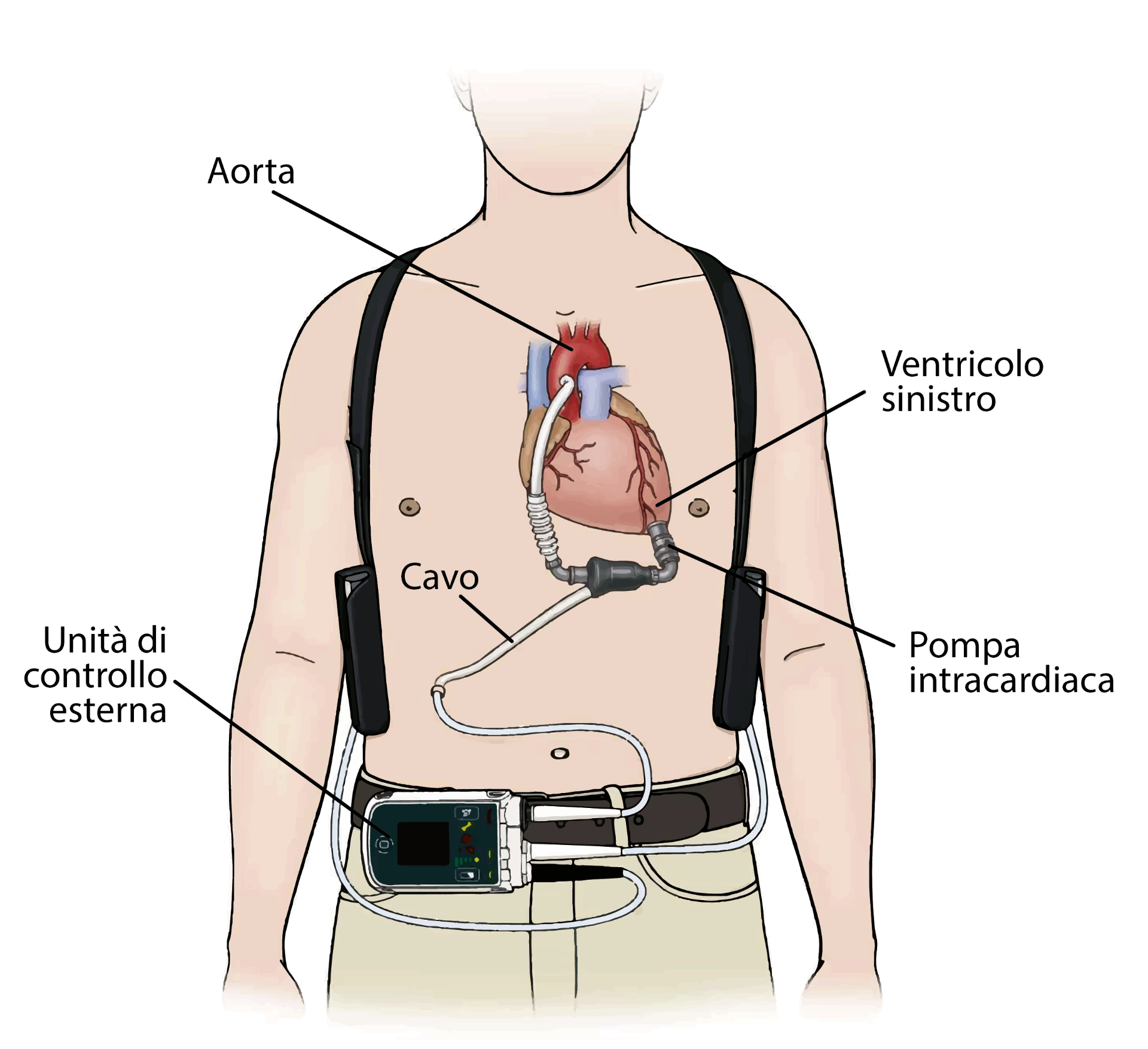
WHAT IS AN LVAD DEVICE
An LVAD (left ventricular assist device) is a mechanical heart pump. When one of the “natural pumps” of the heart (for instance the left ventricle) loses its function due to a pathological degeneration (known as “heart failure”), an LVAD is used to restore the correct hemodynamics of the patient (i.e. the flow of the blood inside the body). The LVAD device includes:
- an intracardiac pump, inserted and connected to the left ventricle
- an external control unit that monitors the pump
- a cable (driveline) that connects the pump to the control unit and supplies power to the pump itself
The pump draws the blood from the ventricle and returns it to the circulation through a cannula inserted into the aorta, thus bypassing the pathological heart and ensuring proper perfusion of the organism.
Illustrative diagram of the LVAD device and its components. Modified from: Left Ventricular Assist Device (LVAD) - Patient information, Golden Jubilee National Hospital, https://www.nhsgoldenjubilee.co.uk/files/4914/9925/3935/LVAD_V3.pdf
Their study focused on thromboembolic complications in LVAD patients, i.e. those complications that develop due to thrombus formation (accumulation of platelets, red blood cells and/or white blood cells) inside the pump. The thrombi hamper or prevent the normal operation of the device; these events are characterized by a high mortality rate. In other cases the thrombus can be “propelled” by the pump itself into the systemic circulation and it reaches small blood vessels (typically the cerebral vessels): this prevents proper blood circulation in the affected vessel and can cause a stroke.
Filippo Consolo states: “By analyzing the data in the literature we have observed that thromboembolic complications in LVAD patients often occur as a consequence of intraventricular thrombus formation, i.e. the thrombus is originally formed inside the ventricle and from there it moves inside the pump. The fact that in these patients the thrombus generates precisely in the anatomical implantation site of the device has led us to suppose the presence of peculiar mechanisms (not entirely known) in that district, that favor the onset of the pathology”.
THE PROJECT
The funded project set out to analyze the cellular mechanisms of interaction between platelets and the endothelium (internal cell lining) of the ventricle.
The researchers used an experimental platform through which they reproduced:
- the mechanical stimulation that the platelets experience when passing through the LVAD
- the pro-inflammatory state of the ventricular endothelium caused, according to literature data, by a high concentration of inflammatory cytokines (molecules) in the blood of LVAD patients.
The two mechanisms were first characterized individually; after that, the researchers evaluated the synergistic effect of the two phenomena.
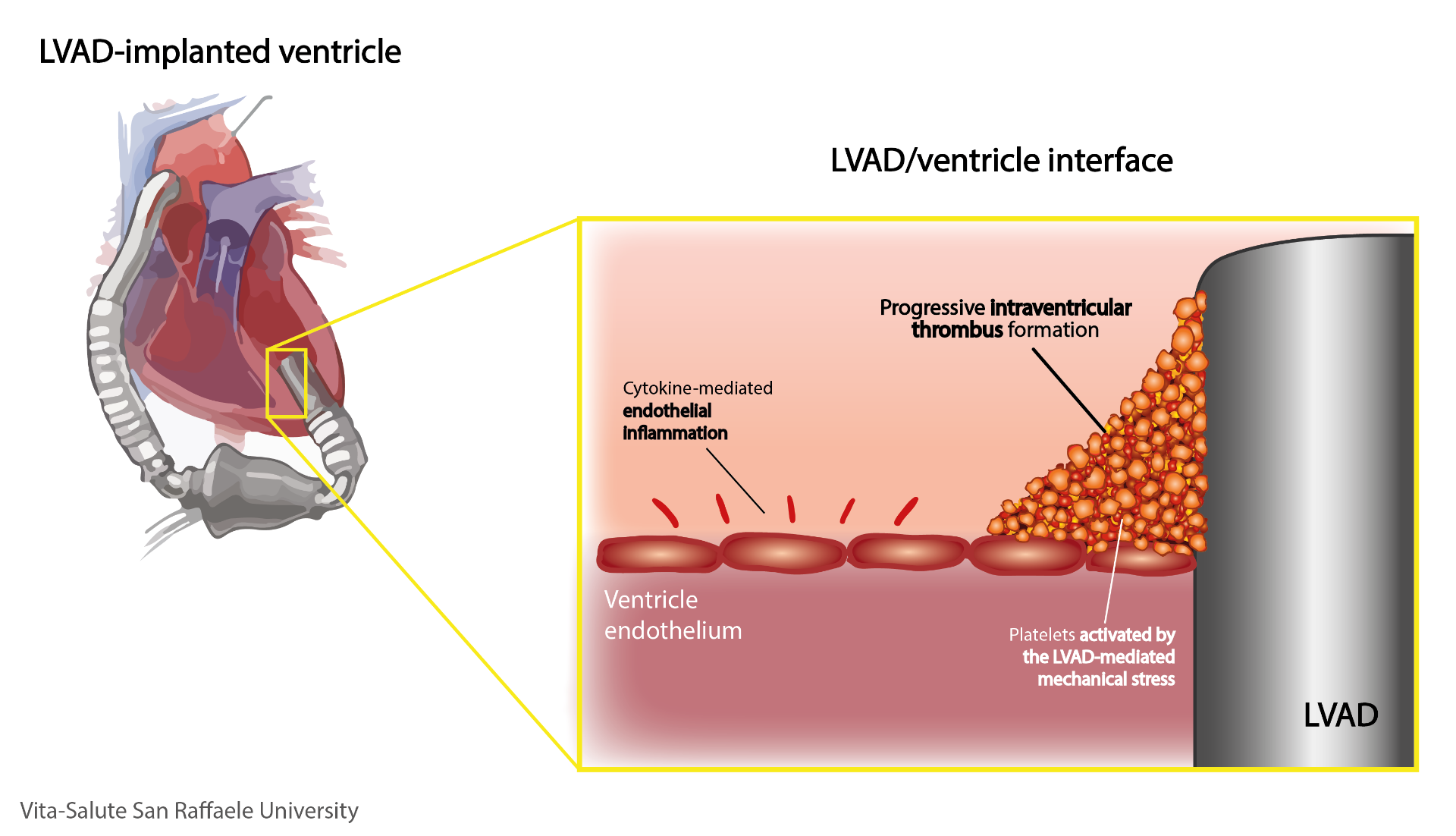
Filippo explains: “According to our results, published in The Journal of Heart and Lung Transplantation, there is a synergy between the two mechanisms that determines a sort of amplification of the pro-thrombotic phenomenon [which favors the formation of thrombi, Editor’s note]: when platelets, activated by the pump-induced shear stress accumulate in the ventricle and interact with the inflamed endothelium, they increase their pro-thrombotic potential. As a result, the platelets adhere significantly to the inflamed endothelium, and begin to express more pro-thrombotic factors than they would if exposed to an uninflamed endothelium. This study specifically characterized for the first time the interaction between the LVAD-induced platelets mechanical activation and the inflammation of endothelial cells”.
The multidisciplinary team (which involved biologists, biomedical engineers, clinical experts in the LVAD sector, cardiologists, intensive care physicians) therefore identified a possible “triggering” mechanism of intraventricular thrombosis, due to a combined effect of LVAD-mediated platelet activation and cytokine-mediated inflammatory endothelium activation.
FUTURE PERSPECTIVES
“The integration of expertise and specific know-how of different disciplines (medicine, bioengineering, biology, biotechnology) has made it possible to reach this goal, otherwise impossible”, says Filippo. “The next step is the identification of a pharmacological therapy aimed at reducing the pro-thrombotic effect associated with inflammation of the endothelium and/or inhibiting the interaction mechanisms (adhesion) between the two cell types that amplify the risk of thrombosis”.
“To date, the antithrombotic therapy used in LVAD patients does not contemplate these phenomena and it is not able to prevent them. Achieving this goal could lead to a significant reduction in the rate of thrombo-embolic complications – often deadly or otherwise highly disabling. Let’s not forget that the LVAD implant represents the only therapeutic option for these patients: their survival is linked to the correct functioning of the device and it is strongly compromised by thromboembolic complications. Reducing complications is therefore essential in order to increase patients’ life expectancy”.
You might be interested in

QS Rankings by Subject 2026

UniSR ranked 1st in Italy for Research and Teaching
/resolutions/res-c660x528/Veschetti_Cariplo_P.-Aeruginosa_UniSR-(1).jpg)
Uncovering the hidden role of bacterial microRNAs in chronic respiratory diseases